Thermal Effects on Ionic Transport and Solid-State Battery Design with a New Blended Polymer Electrolyte: 98[70PEO:30NaCl] +2PVP
1Department of Physics, Government Naveen College, Saragaon, Chhattisgarh, India
2Department of Physics, Bharti Vishwavidyalaya, Durg, Chhattisgarh, India
3Department of Chemistry, Government M.M.R. P.G. College, Champa, Chhattisgarh, India
*Corresponding author E-mail: chandrassi@gmail.com
DOI : http://dx.doi.org/10.13005/msri/220205
ABSTRACT:This study focuses on the ion transport behavior and the fabrication of a solid-state battery using a newly developed sodium ion-conducting blended solid polymer electrolyte (BSPE). The material was prepared with the composition 98 wt.% of [70PEO:30NaCl] blended with 2 wt.% PVP, synthesized via a hot-press technique. Among the tested formulations, this composition demonstrated the maximum ionic conductivity, approximately 3.6×10-5 S·cm⁻¹ at ambient temperature. To understand the ion conduction mechanism across varying temperatures, key parameters such as ionic conductivity (σ), ion mobility (μ), charge carrier density (n), ionic transference number (tₒₙ), and drift velocity (vd) were systematically measured. Activation energies, including migration energy (Em), formation energy (Ef), and drift energy (Ed), were determined using Arrhenius analysis. Utilizing the optimized BSPE composition, a solid-state polymer battery was assembled, and its performance was evaluated under multiple load conditions at room temperature.
KEYWORDS:Activation energy; Blended solid polymer electrolytes; Drift energy (Ed); Migration energy (Em); Formation energy (Ef)
Introduction
The increasing global demand for efficient and safe energy storage systems has intensified research into alternative battery technologies beyond conventional liquid electrolyte-based lithium-ion batteries. Among these, solid-state batteries (SSBs) have emerged as promising candidates due to their enhanced safety, thermal stability, mechanical integrity, and potential for higher energy density.1 The core component of an SSB is the solid electrolyte, which replaces flammable liquid electrolytes and offers better compatibility with lithium metal anodes, leading to safer and more compact battery designs.2
Within the domain of solid electrolytes, solid polymer electrolytes (SPEs) have gained significant attention because of their processability, flexibility, and electrochemical compatibility with lithium salts. SPEs consist of a host polymer matrix typically polar polymers such as polyethylene oxide (PEO) and a dissolved lithium salt like LiTFSI (lithium bis(trifluoromethanesulfonyl)imide). The ionic conduction mechanism in these materials is fundamentally tied to the segmental motion of the polymer chains and the availability of free charge carriers. However, one major limitation of conventional SPEs is their low ionic conductivity at room temperature, typically in the range of 10⁻⁷ to 10⁻⁶ S/cm, which is insufficient for practical battery applications.3
To overcome this limitation, various strategies have been employed, such as polymer blending, plasticization, inorganic filler incorporation, and copolymerization. Among these, polymer blending is an efficient and economical route to enhance ionic conductivity and mechanical strength simultaneously. Blending allows for the tailoring of free volume, amorphous content, and dielectric constant, which are critical parameters for ion transport.4 For instance, blending high ionic conductivity polymers like PEO with mechanically strong but less conductive polymers like poly(methyl methacrylate) (PMMA) or polyvinylidene fluoride (PVDF) can result in optimized systems that balance conductivity, flexibility and structural integrity.5
Temperature is a critical factor influencing ionic conductivity in polymer electrolytes. An increase in temperature generally enhances polymer chain mobility and segmental dynamics, facilitating the migration of lithium ions through the matrix. As temperature rises, the ion pair dissociation increases, leading to a higher number of free charge carriers, thereby improving ionic conductivity. This temperature dependence follows the Arrhenius or Vogel–Tammann–Fulcher (VTF) behavior, depending on the degree of crystallinity and segmental motion of the polymer host.6 Hence, a thorough understanding of the temperature-dependent ionic parameters such as ionic conductivity, activation energy and transference number is essential for designing thermally stable and high-performance polymer electrolytes.
This research presents the formulation and thorough investigation of sodium (Na⁺) ion-conducting blended solid polymer electrolytes (BSPEs) based on the composition: (1–x) [90PEO:30NaCl] + xPVP, with x ranging from 0 to 15 wt%. The ion transport mechanism within these electrolytes was elucidated through a systematic assessment of key electrochemical parameters, including ionic conductivity (σ), ion mobility (μ), mobile charge carrier concentration (n), ionic transference number (tₒₙ), and drift velocity (vd). The composition exhibiting the optimum ionic conductivity was further employed for the fabrication of polymer batteries. The electrochemical performance of the assembled cells was subsequently investigated at room temperature.
Materials and Methods
For the preparation of the present BSPEs: (1–x)[90PEO:30NaCl] + xPVP, where x ranges from 0 to 15 wt.%, analytical-grade poly(ethylene oxide) (PEO; Mw ≈ 10⁵, Aldrich, USA), sodium chloride (NaCl ≥98% purity, Merck, India), and polyvinylpyrrolidone (PVP ≥98% purity, Merck, India) were employed as precursor materials. In prior work, the (70PEO:30NaCl) composition, exhibiting an ionic conductivity of 4.6×10⁻⁶ S·cm⁻¹, was identified and utilized as the host polymer matrix for the preparation of the present BSPE systems. The synthesis involved initially dry-mixing the SPE host and PVP powders in specified weight ratios for approximately 30 minutes at ambient conditions. This mixture was subsequently heated to about 70 °C with continuous agitation for an additional 30 minutes, producing a homogeneous soft slurry. The resulting slurry was then subjected to uniaxial hot-pressing (~1.25 ton·cm-2) between two stainless-steel-cooled plates, forming uniform films with an average thickness of ~0.017 cm. The ion transport behavior in these systems was systematically analyzed through measurements of ionic conductivity (σ), ion mobility (μ), charge carrier density (n), ion transference number (tₒₙ), and drift velocity (vd). Detailed methodologies for these measurements are described in our earlier studies.7–9 Solid-state polymer batteries were subsequently assembled utilizing the optimized BSPE compositions, with cell configurations: [anode//BSPE OCC// cathode]
The solid-state polymer batteries were fabricated using the optimized BSPE composition as the electrolyte separator. The cells were assembled in a conventional configuration, consisting of a sodium-metal anode, the prepared BSPE film as the electrolyte and separator, and a suitable cathode material. The assembly was carried out under an inert atmosphere to minimize moisture and contamination. The electrochemical performance of the cells was examined at room temperature under various external load conditions. The discharge characteristics, including the cell potential as a function of time, were recorded to evaluate the stability and energy delivery capacity of the battery system. These measurements facilitated the assessment of the practical applicability of the synthesized BSPEs for solid-state sodium-ion energy storage devices.
Results
Figure 1 presents the ‘log σ versus 1/T’ relationship for the optimized conducting composition (OCC) of the BSPE: 98(70PEO:30NaCl) + 2PVP. The plot exhibits a linear trend, with a noticeable change in slope occurring near 65–70 °C, which corresponds to the well-documented semicrystalline-to-amorphous phase transition of PEO. Below this transition region, the linear portion of the curve can be accurately described using the Arrhenius expression:
σ (T) = 2.3×10-1 exp (-0.22/ kT) (1)
The activation energy of BSPE OCC is found to be 0.22 eV. For a clearer perspective, the values of key parameters are summarized in Table 1.
Figure 2 shows the differential scanning calorimetric (DSC) thermograms of pure PEO, SPE host: (70PEO:30NaCl) and BSPE OCC: 98 (70PEO:30NaCl) + 2 PVP.
Figure 3 shows the current vs time plot for BSPE OCC: 98 (70PEO:30NaCl) + 2 PVP at room temperature and above. The transport number (tion) obtained is ≈ 0.95, indicating that ~95% of charge carriers are Na⁺ cations with only ~5% contribution from Cl⁻ anions.
Figure 4 illustrates the ‘log μ – 1/T’ and ‘log n – 1/T’ plots for the optimized BSPE: 98(70PEO:30NaCl) + 2PVP. The linear regions of these plots conform to Arrhenius-type behavior and can be expressed as:
μ (T) = 3.8×10 exp (- 0.28/ kT) (2)
n (T) = 5.4×1019exp (- 0.40/ kT) (3)
Here, the values 0.28 eV and 0.40 eV correspond to the migration energy (Eₘ) and formation energy (Ef), respectively. The ionic drift velocity (vd) at different temperatures was determined using the measured IT & n data. Figure 5 presents ‘log vd – 1/T’ plot for the optimized BSPE: 98(70PEO:30NaCl) + 2PVP. The plot demonstrates a clear increase in vd with temperature, and the behavior can be described by the following Arrhenius-type relationship:
vd (T) = 6.5 × 107 exp (- 0.28/ kT) (4)
The value 0.28 eV corresponds to the drift energy (Ed).
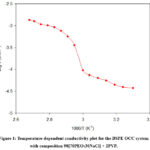 |
Figure 1: Temperature-dependent conductivity plot for the BSPE OCC system with composition 98[70PEO:30NaCl] + 2PVP. |
Table 1: Some important ionic parameters.
| Film | σ (S/cm) | μ(cm2 V-1 s-1) | n (cm-3) | tion | Ea (eV) |
| Pure: PEO | 3.2 × 10-9 | – | – | – | – |
| SPE host: (70PEO:30NaCl) | 4.5 × 10-6 | 7.94 × 10-3 | 9.77 × 1014 | 0.95 | 0.37 |
| BSPE OCC: 98(70PEO:30NaCl)+2 PVP | 3.7 × 10-5 | 3.30 × 10-2 | 7.00 × 1015 | 0.95 | 0.21 |
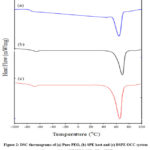 |
Figure 2: DSC thermograms of (a) Pure PEO, (b) SPE host and (c) BSPE OCC system with composition 98[70PEO:30NaCl] + 2PVP. |
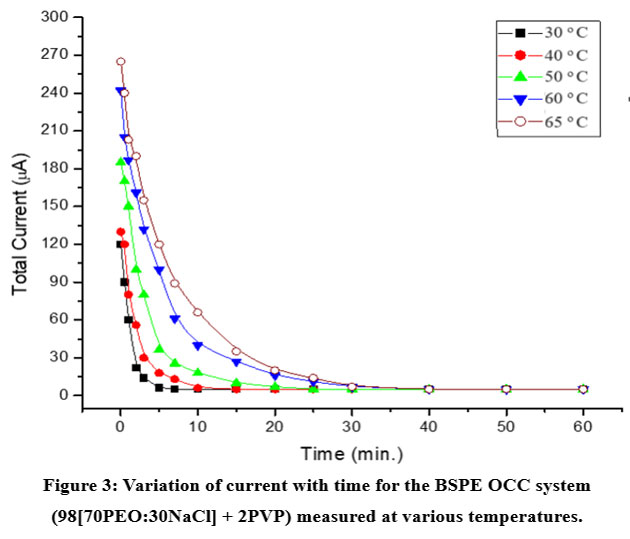 |
Figure 3: Variation of current with time for the BSPE OCC system (98[70PEO:30NaCl] + 2PVP) measured at various temperatures. |
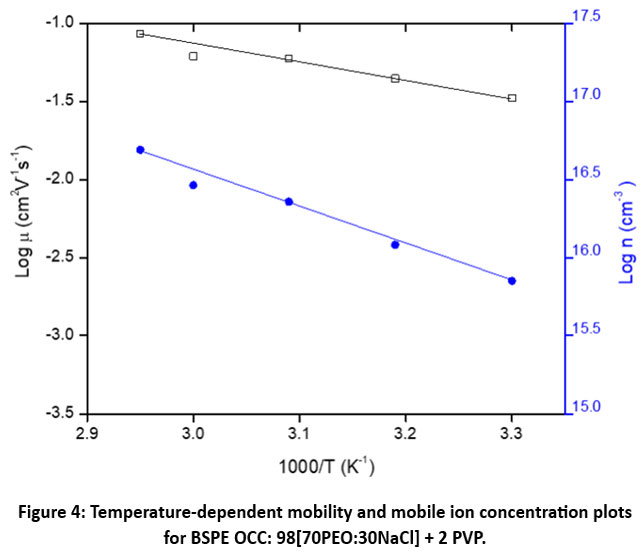 |
Figure 4: Temperature-dependent mobility and mobile ion concentration plots for BSPE OCC: 98[70PEO:30NaCl] + 2 PVP. |
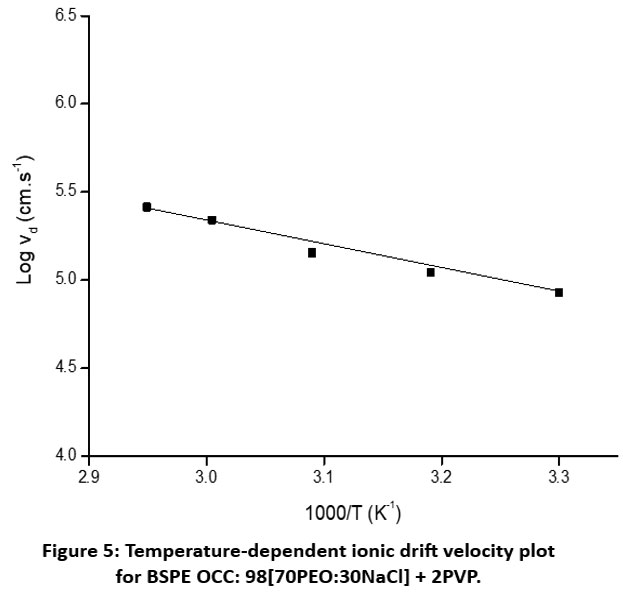 |
Figure 5: Temperature-dependent ionic drift velocity plot for BSPE OCC: 98[70PEO:30NaCl] + 2PVP. |
Figure 5 illustrates the discharge profiles of the solid-state polymer cell at room temperature under two different load conditions: 100 kΩ and 50 kΩ. An open-circuit voltage (OCV) of approximately 3.0 V was recorded for both configurations. Table 2 summarizes key performance parameters of the polymeric battery under different load conditions.
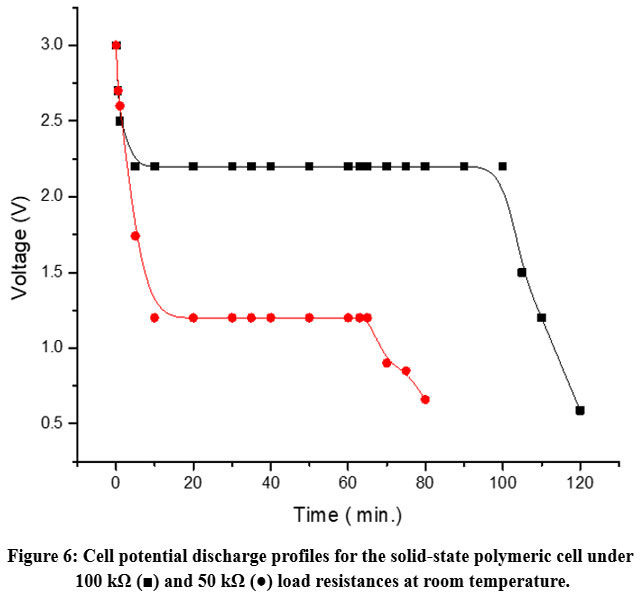 |
Figure 6: Cell potential discharge profiles for the solid-state polymeric cell under 100 kΩ (■) and 50 kΩ (●) load resistances at room temperature. |
Table 2: Key performance parameters of the solid-state polymer cell measured at ambient temperature.
| Load(kΩ) | Working Voltage(V) | Current Density(μA.cm-2) | Discharge Capacity(μA.h) | Power Density(mW.kg-1) | Energy Density(mWh.kg-1) |
| 10050 | 2.21.2 | 1.570.9 | 12133 | 32.0710.39 | 2467415.3 |
Discussion
The observed change in slope in Figure 1 near 65–70 °C confirms the semicrystalline-to-amorphous transition of PEO, which facilitates better ionic conduction. The activation energy (0.22 eV) is slightly lower than that of pure SPE, suggesting enhanced ionic mobility due to increased amorphous content after blending with PVP. This aligns with earlier reports.10,11
It can be noticed from both the thermogrames that the broad endothermic peak was observed in polymeric films at ~ 60-70 oC and it is corresponding to the melting point temperature of pure PEO, as shown in Fig. 2. It is clearly shown from the figure that both the melting temperature (Tm) and glass transition temperature (Tg) have decreased from 70 to 63 oC and -65 to -71 oC respectively. The decrease in Tg is due to the increase in degree of amorphicity and it is directly supports polymer-salt complexation and the increase in ionic conductivity. This type of similar behaviour has been reported in the literature.12-14
The high cationic transport number (t⁺ ≈ 0.95) from Figure 3 indicates that the majority of charge carriers are Na⁺ ions, making the electrolyte highly suitable for sodium-based electrochemical devices.
The temperature dependence of μ, n, and vd (Figures 4 and 5) confirms that conduction is thermally activated. The comparable energy barriers for mobility (0.28 eV) and drift velocity (0.28 eV) indicate that both properties are influenced by similar ionic migration mechanisms. The higher formation energy (0.40 eV) for ions suggests that generating additional mobile species requires more energy than enabling their movement.
The discharge behavior (Figure 6) demonstrates excellent electrochemical stability under low-current drain conditions, with minimal voltage drop over extended durations. This performance highlights the potential of the developed BSPE OCC for practical solid-state polymer battery applications.
Conclusion
This investigation confirms that the synthesized BSPE: 98(70PEO:30NaCl) + 2PVP, exhibits enhanced ionic transport properties at elevated temperatures, underscoring its potential as a viable electrolyte material for solid-state sodium-ion battery systems. The comprehensive evaluation of temperature-dependent ionic parameters including ionic conductivity, transference number, and activation energy reveals enhanced ion mobility and stability at elevated temperatures. Furthermore, the successful fabrication and operation of a prototype solid-state battery validate the practical applicability of the developed BSPE in real-world energy storage systems. These findings contribute meaningful insights toward the development of thermally stable, electrochemically efficient, and safer polymer-based electrolytes, thereby advancing the field of next-generation solid-state energy devices for high-performance and sustainable energy solutions.
Acknowledgment
The author would like to thank Guru Ghasidas Central University, Bilaspur, for providing the experimental facilities.
Funding Sources
The author received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Author Contributions
Angesh Chandra: conceptualization, methodology
Alok Bhatt: final approval of the manuscript
Lumesheari Sahu: data collection, analysis
Archana Chandra: analysis, writing
References
- Goodenough JB, Kim Y. Challenges for rechargeable batteries. J Power Sources. 2010;195(9):3703-3710. doi: 10.1016/j.jpowsour.2009.11.053
CrossRef - Janek J, Zeier WG. A solid future for battery development. Nat Energy. 2016; 1:16141. doi:10.1038/nenergy.2016.141
CrossRef - Armand M, Tarascon JM. Building better batteries. Nature. 2008;451(7179):652-657. doi:10.1038/451652a
CrossRef - Mohan VM, Raja VS, Reddy MJ. Polymer electrolytes for lithium ion batteries: Recent advances and future prospects. J Mater Chem A. 2019;7(20):12289-12303. doi:10.1039/C9TA01788G
- Pandey GP, Hashmi SA. Ionic liquid incorporated polymer electrolytes for lithium battery applications: Ionic conductivity and electrochemical properties. J Power Sources. 2013;237:393-400. doi:10.1016/j.jpowsour.2013.03.094
CrossRef - Zhang H, Ma Y, Wang Y. Temperature dependence of ionic conductivity in solid polymer electrolytes. Solid State Ionics. 2014;268:110-114. doi:10.1016/j.ssi.2014.09.004
CrossRef - Chandra A, Chandra A, Dhundhel RS, Bhatt A. Sodium ion conducting solid polymer electrolyte: temperature dependent ionic parameters and solid-state polymer battery fabrication. Indian J Phys. 2022;96:1069-1074. doi:10.1007/s12648-021-02032-1
CrossRef - Sahu L, Bhatt A, Chandra A, Chandra A. Hot-pressed sodium ion conducting solid polymer electrolytes: Preparation and materials characterization. Mater Today Proc. 2023. In press. doi:10.1016/j.matpr.2023.05.204
CrossRef - Sharma R, Verma A, Chandra A, Chandra A. Temperature dependent ionic parameter studies of a newly synthesized Na⁺ ion conducting nanocomposite polymer electrolytes. Mater Today Proc. 2022;59:15-17. doi:10.1016/j.matpr.2021.09.474
CrossRef - Chandra A. Synthesis and dielectric studies on PEO-PVP blended solid polymer electrolytes. Indian J Pure Appl Phys. 2013;51:788-791. http://hdl.handle.net/123456789/22882
- Dam T, Karan NK, Thomas R, Pradhan DK, Katiyar RS. Observation of ionic transport and ion-coordinated segmental motions in composite (polymer-salt-clay) solid polymer electrolyte. Ionics. 2015;21:401-410. doi:10.1007/s11581-014-1181-5
CrossRef - Chandra A. Hot-pressed PEO-PVP blended solid polymer electrolytes: Ion transport and battery application. Polymer Bulletin. 2016;73:2707-2718. doi.org/10.1007/s00289-016-1616-4
CrossRef - Wu H, Lin J, Zhang Q, Fu Q. Co-continuous polymer blends: morphology control and property design. Adv Mater. 2020;32(14):1906124. doi:10.1002/adma.201906124
- Singh R, Kumar R, Saini P. Tailoring the properties of polymer blends for advanced applications: recent progress and challenges. Prog Polym Sci. 2022;128:101525. doi:10.1016/j.progpolymsci. 2022.101525












