Enhancement of Water Hyacinth Fibre Quality through Sodium Hydroxide Treatment: Part 2.
1Civil Engineering Department, Abubakar Tafawa Balewa University, Bauchi, Bauchi State, Nigeria.
2Building and Construction Department, University of Jos. Plateau State, Nigeria
3Chemical Engineering Department Abubakar Tafawa Balewa University, Bauchi, Bauchi State, Nigeria
Corresponding Author E-mail: auelinwa@gmail.com
DOI : http://dx.doi.org/10.13005/msri/220108
ABSTRACT:The water hyacinth fibre plant (Part II) was carried out using sodium hydroxide (NaOH) solution to improve the quality of the results of the untreated fibre (Part I). The same methods of investigations and tests carried out on the untreated water hyacinth fibre samples were applied to the alkali-treated fibre. The application of the alkali-treatment was done using dilutions of 2, 4, 6, 8, and 10 % NaOH. The treatment of the water hyacinth fibres at 10 % sodium hydroxide was optimum and amorphous with no defined peaks. The basic characteristics of the water hyacinth fibres such as physical and mechanical properties were improved, confirming the effectiveness of the treatment method. The morphology and structural characteristics after treatment showed significant improvement when compared to the untreated water hyacinth fibres. This was due to the removal of the impurities in the lignin, cellulose and hemicellulose. The thermal decomposition process and functional groups of the water hyacinth fibres after treatment have distinct derivative thermogravimetry peak (cellulose) and a high-temperature tail (lignin), and the functional group change of each treatment, respectively.
KEYWORDS:Functional groups; Morphology and structural characteristics; Morphology and structural characteristics; Sodium hydroxide treatment; Thermal decomposition and functional groups; Water hyacinth fibre
Introduction
The preservation of the environment is the principal feature of sustainable development whose impact is by reducing the Green House Gas (GHG). Sustainable development using lignocellulosic in the construction industry therefore justifies the reasons that have been advanced by researchers for replacing synthetic fibres with natural fibres. Lignocellulosic fibres are plant based and have advantages and disadvantages. Some of the advantages are sustainability, consuming less energy, biodegradable and bio-building materials, environmentally friendly, and inexpensive. They have also proven to have both viability and marketability as bio-based building materials 1, 2, and 3. Some of the disadvantages which have great impact on the use of the fibre are its hydrophilic behaviour, intrinsic variability arising from harvesting time and weather variation, etc. Hydrophilic behaviour has been identified as hindering fibre-to-matrix adhesion 4. The cross-section shape and size are important factors which vary from fibre to fibre, and along the length of a single fibre 5. Taj et al 6 also cited poor compatibility, lower durability, and moisture as some of the disadvantages. These disadvantages can be addressed using modern methods of analysis to characterise the chemical and microstructural changes thus enhancing the understanding of the structure-function relationship of WHF 7. These are critical for successful incorporation of WH fibre in concrete production and unlocking vital information on their characteristics and composition.
Rezania et al 8 worked on the composition and structure of water hyacinth stressing the fact that the deconstruction of cellulose, hemicellulose and lignin has varying effects on lignocellulosic biomass. Their findings showed that increasing the cellulose and hemicellulose content, and reducing the lignin content, effectively disrupted the crystalline structure and enhanced the digestibility of WH. The possibility of using dilute sulphuric acid for the treatment of water hyacinth using the FTIR and XRD methods of analysis was investigated by Boonthin et al 9. Their results showed that dilute-sulphuric acid modified the chemical structure of WH. Biswas et al 10 compared the effects of using alkali, acid and hydrogen peroxide pretreatment on the saccharification of WH cellulose. Some of the results showed that the highest sugar content observed was with 5 % NaOH-treated WH. Abral et al 11 reported on the properties of microscale WHF pulp filled with tapioca starch biopolymer composites. They used casting methods to make sample films of bio-composites. Using SEM for the tensile samples, the fracture surfaces displayed good fibre bonding between WH and tapioca starch biopolymer (TSB). The study by Stanislas et al 12 was on okra and raffia fibres. They evaluated the morphology, physio-chemical, and mechanical properties of the materials by subjecting the fibres to four (4) and six (6) wet-dry treatment cycles. This was to assess the effect of the number of cycles in the performance of each fibre. The results showed that the density increased while the fibre lumas the cycles increased, and the statistical analysis showed significant improvement in elastic modulus and tensile strength of raffia fibre. However, there was no effect on the okra fibre.
The present study is on the treatment of WH plant fibre in dilutions of sodium hydroxide (NaOH) solution (2-10 %), at intervals of 2 % increment, and subjecting them to the same test procedures as in Part I of this work for comparisons.
Materials and Methods
Water hyacinth plant fibre characterization 7 was extensively treated in the Part I of this study and will not be repeated here. Part II is an improvement (treatment) work using sodium hydroxide dilutions (2-10 %) at increment of 2 %. The treatment was carried out using forty (40) grammes of the WHFs and submerging them in one litre solutions of NaOH for the stipulated dilutions for three (3) hours in a water bath at room temperature. After the appropriate soaking time the water hyacinth fibres were drained and rinsed thoroughly and neutralized with diluted acetic acid. The fibres are washed again with distilled water to remove pectin, lignin and any excess NaOH to obtain a neutral pH. The water hyacinth fibres were subsequently spreadout in loose bunches for drying at room temperature for 48 hours which ensured even drying to reduce the effects of clumping allowing for their easy separation without damage. The WHFs are cut into varying lengths of 10, 20, 30, 40 and 50 mm. This is shown in Table 1.
Table 1: Weight of NaOH pellets in grams
| NaOH (%) | Weight of NaOH (g) |
| 2 | 20 |
| 4 | 40 |
| 6 | 60 |
| 8 | 80 |
| 10 | 100 |
The results of the biological and chemical properties of the WHFs tested in accordance with AOAC 13 and TAPPI T264 cm-97 (20000a) 14 are shown in Table 2.
Table 2: Biological and Chemical Characterization of WHF
| Parameter (%) | Untreated 7 | Treated | Difference (%) |
| Cellulose | 14.1 | 16.5 | 17.02 |
| Hemicellulose | 21.5 | 11.2 | – 47.91 |
| Lignin) | 7.1 | 4.9 | – 31.00 |
| Ash | 10.9 | 8.0 | – 26. 61 |
| Crude Protein | 3.9 | 3.5 | – 10.26 |
| Crude Fibre | 29.1 | 33.6 | 15.46 |
The physical properties of the water hyacinth fibres (WHs) were tested in accordance with ASTM D 570-98 standard 15 using dried water hyacinth fibre samples of 10g. These were cut into lengths of 40-50 mm and immersed in water for 24h 7. The excess water was removed with a dipper. The saturated WHF samples were weighed and dried in an oven for 24h. The results are given in Table 3.
Table 3: Physical Properties of WHF
| Parameter | Untreated 7 | Treated | Difference (%) |
| Moisture content (%) | 13.7 | 12.8 | – 6.57 |
| Diameter. (µm) | 320 | 174.3 | – 45.63 |
| Density. (kgm-3 | 666.7 | 1253.3 | 87.99 |
| Specific gravity | 0.857 | 1.458 | 70.13 |
| Water absorption (%) | 1067.0 | 447.0 | – 58.11 |
The elemental composition of the untreated water hyacinth fibre plant was analyzed using the Thermo-Fisher Scientific elemental and radiation detection solution. The elemental composition of the WHFs analyzed results are shown in Table 4.
Table 4: Elemental Composition of Untreated and Treated WHF
| S/No | Element | Symbol | Water Hyacinth Fibre (%) | |
| Untreated | Treated | |||
| 1 | Calcium | Ca | 0.11 | 2.15 |
| 2 | Potassium | K | 2.52 | 0.10 |
| 3 | Chlorine | Cl | 2.51 | 0.03 |
| 4 | Sodium | Na | 0.00 | 0.12 |
| 5 | Magnesium | Mg | 0.03 | 0.03 |
| 6 | Sulphur | S | 0.07 | 0.03 |
| 7 | Nickel | Ni | 0.00 | 0.02 |
| 8 | Phosphuros | P | 0.05 | 0.02 |
| 9 | Iron | Fe | 0.03 | 0.04 |
| 10 | Aluminium | Al | 0.05 | 0.04 |
| 11 | Manganese | Mn | 0.03 | 0.08 |
| 13 | Strotium | Sr | 0.00 | 0.01 |
| 19 | Silicon | Si | 0.01 | 0.11 |
| 20 | Barium | Ba | 0.03 | 0.18 |
| 21 | Tin | Sn | 0.00 | 0.19 |
| 22 | Bismuth | Bi | 0.04 | 0.03 |
| 23 | Lead | Pb | 0.31 | 0.00 |
| 24 | Silver | Ag | 0.00 | -0.01 |
| 25 | Niobium | Nb | 0.00 | 0.01 |
| 27 | Tungsten | W | 0.04 | 0.01 |
The mechanical properties of the WHFs were tested according to ASTM D 3822M-14 16. A gauge length of 60 mm was used and three (3) fibre samples were tested for the various mechanical properties.The results are shown in Tables 5 , 6, 7 and Figure 1.
 |
Figure 1: Mechanical Characteristics of WHF (Untreated/Treated).Click here to View Figure |
Table 5: Mechanical Characteristics of WHF
| Type of Test | Treatment (%)/Value x 10-2 | |||||
| 0.0 7 | 2.0 | 4.0 | 6.0 | 8.0 | 10.0 | |
| Maxm Tensile Stress (MPa) | 1520.00 | ND | ND | ND | ND | 16850.00 |
| Load @ Maxm Tensile Stress (N) | 122.00 | 247.00 | 243.00 | 38.00 | 242.00 | 398.00 |
| Tensile Strain @Maxm Tensile Stress (Mm/Mm) | 2.00 | 2.00 | 1.00 | 2.00 | 1.00 | 4.00 |
| Tensile Extension @ Maxm Tensile Stress (Mm) | 110.00 | 98.00 | 0.68 | 1.35 | 0.78 | 2.23 |
| Energy @ Maxm Tensile Stress (J) | 0.1 | 0.1 | 0.1 | 0.3 | 0.1 | 0.6 |
| Tensile Stress @ Break (Standard) (MPa) | 0.6 | -2.30 | -2.40 | -3.70 | -1.20 | 0.00 |
| Load @ Break (Standard) (N) | 6.00 | -23.00 | -24.00 | -37.00 | -12.00 | -0.4 |
| Tensile Strain At Break (Standard) (Mm/Mm) | 5.00 | 3.00 | 3.00 | 5.00 | 4.00 | 5.00 |
| Tensile Extension @ Break (Mm) | 289.00 | 177.00 | 175.00 | 276.00 | 211.00 | 318.00 |
| Energy At Break (Standard) (J) | 0.01 | 0.1 | 0.1 | 0.3 | 0.1 | 0.6 |
| Modulus (E-Modulus) (GPa) | 770.00 | 2510.00 | 2280.00 | 2480.00 | 2410.00 | 1530.00 |
| Tensile Stress at Yield (Offset 0mm/Mm) (MPa) | ND | 9.80 | 21.70 | 17.00 | 18.50 | 24.50 |
Table 6: Standard Deviation of the Mechanical Strength Characteristics of the WHF
| Test | Unit x 10-2 | NaOH Treatment (%)/Average Value | |||||
| 0 .0 7 | 2.0 | 4-0 | 6.0 | 8-0 | 10.0 | ||
| Max Tensile Stress | Mpa | 7.62 | 12.00 | 4.99 | 3.19 | 8.31 | 27.94 |
| Load @ Maxm Tensile Stress) | N | 76.24 | 119.84 | 49.92 | 31.90 | 83.06 | 279.41 |
| Tensile Strain @ Maxm Tensile Stress | mm/mm | 1.09 | 0.89 | 0.48 | 0.43 | 0.63 | 0.95 |
| Tensile Ext @ Maxm Tensile Stress | mm | 65.39 | 53.46 | 28.87 | 25.98 | 37.53 | 56.87 |
| Energy @ Maxm Tensile Stress | J | 0.06 | 0.09 | 0.06 | 0.04 | 0.08 | 0.55 |
| Tensile Stress At Break (Standard) | MPa | 0.28 | 0.86 | 3.08 | 1.76 | 3.80 | 3.39 |
| Load @ Break (Standard) | N | 2.84 | 8.58 | 30.79 | 17.56 | 38.02 | 33.92 |
| Tensile Strain at Break (Standard) | mm/mm | 1.76 | 0.61 | 0.0050 | 0.0125 | 0.0084 | 0.0048 |
| Tensile Ext @ Break (Standard) | mm | 105.48 | 36.86 | 0.3006 | 0.7496 | 0.5026 | 0.2887 |
| Energy @ Break (Standard) (J) | J | 0.06 | 0.1 | 0.004 | 0.008 | 0.0008 | 0.00569 |
| Modulus (E-Modulus) (Gf/Tex) | *gf/tex (x 105)* | – | 205.95 | 56.65 | 56.51 | 106.64 | 84.73 |
| Tensile Stress at Yield (Offset 0 Mm/Mm) | MPa | – | – | – | – | – | 21.60 |
Table 7: Performance Characteristics of Untreated and Treated WHFs at 10 % NaOH
| Parameter | Fibre Condition | Diff. (%) | |
| Untreated7 | Treated | ||
| Maxm Tensile Stress (MPa) x 10-2 | 1520.00 | 16850.00 | 1009 |
| Load @ Maxm Tensile Stress (N) x 10-2 | 122.00 | 398.00 | 226 |
| Tensile Strain @ Maxm Tensile Stress (Mm/Mm) x 10-2 | 2.00 | 4.00 | 100 |
| Tensile Extension @ Maxm Tensile Stress (Mm) x 10-2 | 110.00 | 2.23 | – 98 |
| Energy @ Maxm Tensile Stress (J) x 10-2 | 0.1 | 0.6 | 500 |
| Modulus (E-Modulus) (GPa) x 106 | 770.00 | 1530.00 | 99 |
X-Ray Diffraction Analysis of WH Fibre
Water hyacinth plant composite structural X-ray diffraction patterns were identified with the help of the X-ray diffraction method. Conventionally, this method was used to recognize the amorphous and crystalline phases of the material. The amorphous and crystalline phases of the WHFs were determined using XRD analysis method model radiation at operating conditions of V = 45VA, I = 40mA diffractogram shown in Figure 2. The structure of the produced treated and untreated water hyacinth fibres and their associated properties were determined by their cellulosic and non-cellulosic composition in the natural or treated state of the fibre, and to a large extent they determine the degree of crystallinity of the fibre. Figure 2 depicts the XRD patterns of NaOH treated and untreated water hyacinth fibres. The figure confirmed the identification of peaks for both water hyacinth fibre samples at 2 range of 5 – 80o. The crystallinity index (CI) is used to measure the fractional part of the fibre material in the crystalline phase. The CI of the treated and untreated water hyacinth fibres produced were determined using Equation 1 as follows:
![]()
Where ICP is the peak intensity of the more crystalline peak, IAP is the peak intensity of the broad and amorphous peak. Crystallinity shows the extent of long-range orderliness in the matrix configuration of a material. The more crystalline a fibre is, the better aligned or ordered are its chains and thus, the potential for enhancement of compressive and flexural strength of concrete.
 |
Figure 2: X-ray Diffractogram of Untreated and Treated WHF.Click here to View Figure |
The thermogravimetric analysis (TGA/DTA) monitored the changes in the weight loss and thermal stability, subjecting the samples to steady increase of temperature 17.. The results are shown in Figure 3a and Figure 3b, and Figure 4a and Figure 4b).
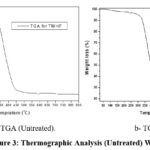 |
Figure 3: Thermographic Analysis (Untreated) WHF.Click here to View Figure |
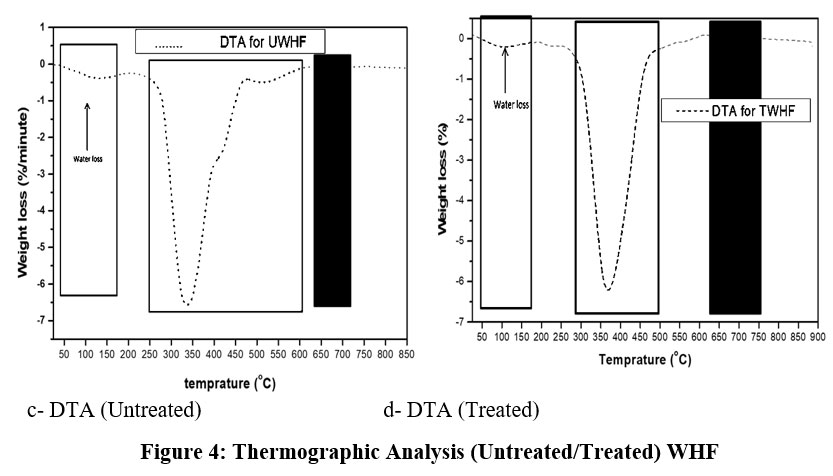 |
Figure 4: Thermographic Analysis (Untreated/Treated) WHF.Click here to View Figure |
The Fourier transform infrared spectroscopy (FTIR) was used to investigate the changes The changes of the functional groups of the WHFs surfaces were studied using the FTIR method of analysis. This converted the raw data from the broad-band source to obtain the absorbance level at each wavelength. The results of the untreated WHF 7 and treated are shown in Figures 5 and Figure 6, respectively.
 |
Figure 5: FTIR Spectroscopy of Untreated WHF.Click here to View Figure |
 |
Figure 6: FTIR Spectroscopy of Treated WHFClick here to View Figure |
The surface morphology of the WHFs was investigated using the SEM method of analysis and the results are shown in Figure 7a and Figure b. Table 8 is a comparison table for the structural characteristics of the Untreated and NaOH Treated WHF.
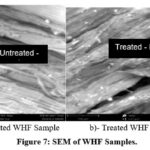 |
Figure 7: SEM of WHF Samples.Click here to View Figure |
 
Table 8: Elemental Composition of Untreated and Treated WHFs
| Element | Symbol | Weight Composition (%) | Difference (%) | |
| Untreated 7 | Treated | |||
| Calcium | Ca | 31.86 | 35.36 | 11.00 |
| Potassium | K | 26.02 | 7.87 | – 69.80 |
| Chlorine | Cl | 17.53 | 5.55 | – 68.34 |
| Carbon | C | 4.52 | 3.22 | – 27.76 |
| Aluminum | Al | 3.58 | 2.65 | – 25.98 |
| Silicon | Si | 3.42 | 5.79 | 69.30 |
| Iron | Fe | 2.73 | 3.28 | 20.15 |
| Sulphur | S | 1.88 | 3.01 | 60.11 |
| Phosphorous | P | 1.42 | 2.04 | 43.66 |
| Magnesium | Mg | 1.41 | 2.17 | 53.19 |
| Sodium | Na | 1.38 | 10.69 | 674.64 |
| Nitrogen | N | 1.03 | 0.53 | – 48.54 |
Results and Discussions
Biological, Chemical and Physical Characteristics of Water Hyacinth
Table 2 showed an appreciable increase for the cellulose (17 %) and crude fibre (15.5 %) after the NaOH-fibre treatment, while the hemicellulose, lignin, ash and crude protein approximately decreased by 48 %, 31 %, 27 %, and 10 % respectively. The effect on the cellulose, hemicellulose and lignin have been linked to the percentage dilution of NaOH used, and the extractive process 18. However, there are varied opinions on this assertion. Some researchers have recorded an increase on the cellulose 8, 10 and 19, and on hemicellulose 8, 19, while decreases were reported on hemicellulos 8, and lignin 8. 10, 19, respectively. The treatment of WH with NaOH caused the lignin molecules to degrade, and the lignocellulosic biomass to swell, thereby increasing the internal surface area, decreasing the degree of crystallinity, and disruption of the lignin structure. The increase in cellulose, attributed to decrease in lignin, was because of its solubilization in an alkali aqueous solution. This reduced the lignin in biomass, increased the surface area and allowed the penetration of water molecules to inner layers, and broke the bonds between lignin and hemicellulose carbohydrate 20. Thus, alkali pretreatment can be said to be an efficient method for removing the lignocellulosic biomass 21. The cell wall of WH plant as reported by Singh et al 22 was exposed to complex heterogenous interactions of lignin, hemicellulose, and cellulose, and prevented the cell wall from being inaccessible to microorganism and enzymes during their facile bioconversion to sugar and other intermediates of high yield. Therefore, Brandt et al 23 held the opinion that in untreated biomass the polymers of cellulose, hemicellulose, and lignin are linked as strong structure, and the compositional analysis of these components after treatment showed how they had been removed or changed. Thus, the removal of some recalcitrant compounds including various forms of uronic acid, acetyl groups and lignin from lignocellulosic biomass was an essential duty of pretreatment technique 22.
Table 4, the XRF results contained twenty-nine (29) elements. The untreated fibre with wt. % < 0.01 % was approximately 52 % (14), while the alkali treated water hyacinth fibre was 34 % (10) of the total. The implication of this is that NaOH treatment was very effective in improving the characteristics of the material. Calcium (2.5 %) was the dominant while Potassium. (2.5 %) and chlorine (2.5 %) contents were substantially reduced after the treatment to 0.10 % and 0.03 %, respectively.
Figure 1, Tables 5 and 6 are the tensile strengths/the statistical standard deviation characteristics of the untreated and treated WHFs. Table 7 showed the compared performance characteristics of the untreated and NaOH-treated fibre showing that the fibres are greatly improved after treatment.
XRD Analysis of the WHF
From the figure, two broad peaks were observed at 2 values of 14.1o and 21.5o which corresponds to [1 1 0] and [2 0 0] crystal lattice planes of water hyacinth respectively. This was similar to the XRD result obtained from the work of Motaleb et al. 26 on production of biocomposite from water hyacinth and sugar cane bagasse. Two sharp peaks observed from the untreated water hyacinth XRD pattern at 26.1o and 27.5o may be an indication of the presence of a crystalline impurity in the untreated sample. Therefore, they do not represent the peaks for the water hyacinth fibre. The broad peaks of untreated fibre signified the presence of amorphous materials in the fibre such as lignin, pectin, cellulose and/or hemicellulose 27. The broad peaks were observed to be transformed to sharper peaks with higher peak intensities on alkali treatment of water hyacinth fibre using NaOH. This was an indication of the transformation of the fibre from the predominant amorphous phase to a crystalline phase. This finding was attributed to the partial removal of amorphous hemicellulose, wax and other non-crystalline components from the fibre material 28. The crystallinity index of untreated water hyacinth fibre was calculated to be as low as 6.1%, portraying the untreated sample as a highly amorphous material. Treatment with NaOH enhanced the crystallinity of the water hyacinth bio-filler to a CI value of 41.2%. The increment in CI is related to the dissolution and removal of amorphous fraction present in the fibre 29. Crystallinity of fibre is crucial towards its ability to improve the strength of concrete.
TGA/DTA of WHFs
The TGA graphs for the untreated and treated WHFs samples are shown in Figures 3 (a and b) and Figure 4 (a and b), respectively. The fibre samples showed decomposition with respect to temperature from 40-140OC recording weight losses of 15 wt. % and 10 wt. % respectively, for the untreated and NaOH-treated samples, showing that water/moisture evaporated from the samples. The lower values recorded for the treated sample was an indication that some of the hydrophilic functional groups, mostly the extractives and some fractions of hemicellulose, originally present in the sample were removed via the NaOH-treatment8. Noticeable shoulders were observed for the untreated and treated samples from 221-346 ⁰C and 243-363 ⁰C, respectively, with corresponding weight losses of 48 wt % and 42 wt %, respectively, attributed to the decomposition of holocellulose which were mainly hemicellulose and some part of cellulose in the samples. It was evident in the treated sample that some of the lignin that madeup the biomass were reduced during the NaOH-treatmentt stage, and therefore, responsible for the lower percentage weight loss relative to the untreated sample 22. A visible short shoulder was further observed at 350-426 ⁰C which represented the cellulose decomposition. This accounted for about 9.0 wt % for the untreated sample and 6.0 wt % for the treated sample. Lower degradation behaviour, exhibited by the NaOH-treated sample, within this temperature regime, was attributed to the improved crystallinity index 22. The weight loss between 450-700 ⁰C was the residual lignin decomposition. This accounted for 4.5 wt % and 2.5 wt % weight losses, respectively, for the untreated and NaOH-treated samples. Two main peaks for the untreated and NaOH-treated samples were identified around 300 ⁰C and 450 ⁰C, respectively. These are shown in Figure 3b and Figure 4b of the DTG curves, and could be due to early and late degradation of cellulose. The peak at around 300 ⁰C was due to the degradation of hemicellulose with some components of cellulose. Reaction intensities of 1.2 wt%/⁰C and 3.7 wt%/⁰C which corresponded to early and late cellulose decomposition were exhibited by the untreated WHFs sample as shown in Figure 3a. Comparing this with the NaOH-treated samples, the early and late cellulose decomposition spanned over wide range of temperature due to reduction in amorphous components in the sample. The reaction intensities recorded were 0.7 wt%/⁰C and 2.7wt%/⁰C, which suggested fewer complex reactions at both early and late cellulose decomposition stage with probable multiple parallel complex reactions emanating from hemicellulose, cellulose and lignin.
FTIR Spectroscopy Analysis of the WHF
Figure 5 and Figure 6 of the FTIR show the peaks for both the untreated and NaOH-treated WH fibres. The various functional groups in FTIR are shown in Table 9 and ranged from 1028.7 cm-1 to 3366 cm-1, and 1013.8 cm-1 to 3280.1 cm-1, respectively, indicating the existing of lignin, cellulose, and hemicellulose, the main component of WH. Details of the characteristics of the various FTIR functional groups. The bands of NaOH-treated WH absorbed more materials because the O_H group is associated with hydrogen bonding 31. The C-H asymmetrical stretching vibrations of CH2 and CH in the cellulose and hemicellulose components (2918.5 cm-1, untreated), and 2922.2, NaOH-treated), are attributed to carboxyl acid 32, 33, 34. The appearance of a new peak at 2855.1 cm-1 (NaOH-treated), indicated a C-H stretching vibration of CH2 and CH in cellulose and hemicellulose component 34. Peaks of C-H and C-O stretch deformations were observed in both the untreated and treated samples of WHF at 1722.0 cm-1 and 17748.1 cm-1, attributed to the stretching bending of C=O due to the presence of organic compounds and impurities. The region affected by treating the WHF with NaOH is 1748.1 cm-1. The same had been collaborated by Vinod et al 35. The peaks between 1602.8 cm-1 and 1423 cm-1 for the untreated were due to the C=C aromatic skeletal vibration of lignin. The stretching of the alkenes and the reduction in its intensity at Peak 1602.8 cm-1 showed the removal of the lignin. Barki and Jayamani 36, and Vardhini et al 37 confirmed same behaviour working with bamboo fibre. The Peak at 1423.8 cm-1 was due to the C-H bond in the aromatic ring of the lignin showing lower intensities, which indicated the removal of lignin and hemicellulose contents in the fibres. The absence of Peak 1244 cm-1 in the NaOH-treated WHF sample indicated the removal of lignin and hemicellulose. This assertion was similar to the results of Vishnu Vardhini et al 37 who worked on banana fibre samples. The implication was that the treatment made the C-O-C asymmetric stretching thinner with the acetyl group removal of lignin and hemicellulose. The NaOH-treatment reduced the hydrogen bonding because of the disappearance of the hydroxyl group38. The C-O stretching peaks for the untreated WHF and NaOH-treated WHF at 1028.7 cm-1 and 1013.8 cm-1 showed the presence of an alcoholic hydroxyl group, and the intensity reductions showed removal of lignin and hemicellulose from the WHF. The peak 872.2 cm-1 is the C-H stretching of the glycosidic linkages showing reduction due to the treatment. The treated WHF as shown by the FTIR spectrum contained greater percentage of cellulose than lignin or hemicellulose. Therefore, the data suggested that WHF is mainly lignocellulose in composition supported by the fact that the most intense bands in the FTIR (Untreated) were 3366.1cm-1, 2918.5 cm-1and 1028.7 cm-1 and attributed to O-H and C-H stretch vibrations, and typical of cellulose. The treatment of the WHF with 10 % NaOH showed that the presence of the -OH group was significantly reduced, and the increase in peaks confirmed the analysis regarding the removal of some hemicellulose and lignin structures along the fibre surface. Hemicellulose structure is branched and networked unlike cellulose which is mostly a linear polymer, and this network structure did not provide enough -OH groups to be exposed to the surface of the fibres. Lignin was believed to be polycyclic and aromatic with some degree of branching contained even less -OH groups 39. From the peaks in the treated samples, it is evident that cellulose, hemicellulose, and lignin were removed as the complex carbohydrate bonds were broken during the alkaline treatment.
Table 9: Characteristics of the FTIR Spectroscopy Analysis of the WHF
| Wave Number (cm-1) | Stretching Vibrations | Assignment | |
| Untreated | Treated | ||
| 3366.0 | 3280.1 | -OH – Stretching Vibrations | Lignin, Cellulose, Hemicellulose |
| 2918.5 | 2922.2 | C-H (CH2 and CH) Stretching vibrations | Carboxyl Acid |
| 2855.1 | C-H (CH2 and CH) stretching vibrations | Cellulose and Hemicellulose | |
| 1722.0 | 17748.1 | C=O – stretching vibrations | Organic Compounds and Impurities |
| 1748.1 | C=C Aromatic stretching | Lignin | |
| 1602.8-1423.0 | C=C Aromatic Stretching Vibration | Removal of Lignin | |
| 1602.8 | |||
| 1423.8 | C-H Bond in the Aromatic Ring | Removal of lignin and Hemicellulose | |
| 1423.8 | |||
| 1028.7 | 1013.8 | C-O Stretching Vibrations | Alcohol Hydroxyl Group |
| 872.2 | C-H Stretching Vibrations | Glycosidic Linkages | |
Morphological and Structural Characteristics of Untreated and Treated WHFs
Morphological Characteristics of Water Hyacinth Fibre7a)
Figure 7 is the SEM and EDX spectrograms of WHF samples showing the morphology for both untreated WHF samples (Figure 7a) and NaOH-untreated WHF samples (Figure 7b). Significant differences were observed after the NaOH-treatment, and these were due to the removal of the impurities in the untreated WHF samples. The impact of the treatment was healthier and improved morphology and reduced roughness of the fibre surface 19. The interactions of the untreated samples with sodium hydroxide led to the removal of the wax and cuticles on the surfaces of the untreated WHF thereby reducing the size of the diameter. This also removed the superficial layers and increased the contact area. This is advantages for the fibre-matrix adhesion because it facilitates both the mechanical interlocking and bond reaction40. Therefore, the microscopic analysis of fibre surface morphology is of utmost importance in characterizing the structural changes that occurred upon the treatment. This helped in the cleansing and smoothening of the fibres.
Structural characteristics of WHF
Table 8 is the structural characteristics of untreated and NaOH-treated WHF samples, and with twelve (12) elements in both WHF samples with varying wt. % concentrations. The NaOH-treated samples showed an increase of 58.3 % in the structural wt. % of WHF samples , while the untreated WHF samples decreased by 41.7 %. The affected oxides for the NaOH-treated are Ca (11 %), Si (69.30 %), Fe (20.15 %), S (60.11), and Na (674.64 %), while that of the untreated WHF are. P (-69.80 %), Cl (-68.34 %), C (-27.76 %), Al (-25.98 %), and N (-48.54 %), respectively. These results showed that the alkaline treatment of the WHFs was very effective, and thus, improved the surface morphology of the WHFs.
Conclusion
In this work, characterization of untreated and 10 % NaOH-treated WHFs was done by using the XRF, XRD, FTIR, TG/DTA and SEM methods, and the following conclusions are made on the study.
There was significant improvement on the physico-chemical and mechanical properties of the WHF samples. The cellulose and crude fibre increased appreciably, while hemicellulose, lignin, ash and crude protein decreased after the alkaline treatment. The various metal mineral elements in various concentrations in the WHF plant detected using the XRF spectroscopy showed that the alkaline treatment gave improved characteristics to the WHFs because the NaOH solution allowed sodium ions (Na+) to penetrate and interact with the cellulose structure, leading to the incorporation of sodium into the fibre.
The mechanical strength (tensile strength, energy at breaking load, and young modulus) were all improved after the alkaline treatment. The tensile tests on WH fibres reveal that mechanical properties of alkali treated fibres are superior to those of untreated fibres.
The morphology of the untreated WH fibre showed a well-shaped fibril with a rigid lignin structure coated surface with the capacity to hold the liquid contents with good absorbency. The 10 % NaOH treated WHFs showed significant differences in the morphological and structural characteristics compared with the untreated WHFs. The differences were due to the removal of the impurities in the untreated WHFs.
The XRD diffractogram characterization shows that the untreated WH fibre is crystalline in nature, while the alkaline treated WHF is amorphous. The crystallinity index of untreated WH fibre is 6.1 % and the treated with 10 % NaOH is 41.2 %. Therefore, NaOH treatment enhanced the crystallinity of WH fibre, and the increment is related to the dissolution and removal of amorphous fraction present in the fibre.
The FTIR showed the functional group change of each treatment respectively. The FTIR peak analysis of the untreated WHF shows substantial changes in the absorption of cellulose functional groups and the elimination of lignin signals. The TGA measurements of untreated and treated fibres revealed that NaOH treatment had a positive impact on the thermal stability of samples. The thermal decomposition process of untreated WH fibre resulted in similar TG and DTG curves due to being lignin cellulosic material. These curves showed a distinct DTG peak (cellulose) and a high-temperature tail (lignin). The alkali treatment on WH significantly removed surface impurities on fibres, producing modifications on the surface and enhanced its thermal stability. The SEM images show firm, compact, highly ordered, and rigid fibril structures without degradation in the untreated WH plant fibre sample, while the 10 % NaOH treated samples exhibited loose, dispersed, and distorted structures.
Acknowledgment
The authors would like to acknowledge the Department of Civil Engineering, Abubakar Tafawa Balewa University, Bauchi, Bauchi State, Nigeria, for the facilities and access to some of the important equipment used for the investigations.
Funding Sources
The authors received no financial support for the research.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
The manuscript incorporates all datasets produced or examined throughout this research study.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Permission to reproduce material from other sources
Not Applicable
Authors’ Contribution
A.U.Elinwa: Visualization, Supervision, Project Administration, Review and Editing
A.A. Ishaya: Conceptualization, Methodology, Data Collection, Analysis Writing -Original Draft.
I.Y. Mohammed: Analysis, Review and Editing, Supervision
References
- Ajithram A, Jappes JTW, khan MA, Brintha NC. Evaluation of mechanical properties and thermal characteristics of aquatic wastewater hyacinth (Eichhornia crassipes) plant into natural powder and ash reinforced polymer composites for lightweight applications. Proc IMechE Part C: J Mechanical Engineering Science. 2021; 0 (0) 1–12
- Kumar N, Grewa J.S, Kumar S, Kumar N, Kashyap K. Mechanical and thermal properties of NaOH treated sisal natural fiber reinforced polymer composites: Barium sulphate used as filler. Mater. Today: Proc. 2021, 45 (6) 5575–5578, https://doi.org/10.1016/j.matpr.2021.02.30
CrossRef - Kamath S.S, Chandrappa R.K. Additives used in natural fibre reinforced polymer composites-a review. Mater. Today: Proc., 2022, 50 (5) :1417–1424.
CrossRef - Navarro C.I.T, Paciornik S, d’Almeida J.R.M. Microstructural characterization of natural fibers: etlingera elatior, costus comosus, and heliconia bihai, Conference Papers in Materials Science. 2013, Article ID 878014, 7 pages http://dx.doi.org/10.1155/2013/878014
CrossRef - Bergfjord C, Holst B. A procedure for identifying textile bast fibres using microscopy: flax, nettle/ramie, hemp and jute. Ultramicroscopy. 2010, 110 (9), 1192–1197.
CrossRef - Taj S, MunawarA, Khan S. Natural fiber-reinforced polymer composites. Proc. Pakistan Acad. Sci. 2007, 44: 129–144.
CrossRef - Ishaya, A.A., Elinwa, A.U., Mohammed I.Y. Water hyacinth plant fibre characterization. 2024, 11 (9), 1172-11290.
CrossRef - Rezania S, Alizadeh H, Cho Darajeh J.N, Park J, Hastemi B, Din, M.F.M, Krishnan S,. Yadar K.K, Gupta N, Kumar S. Changes in composition and structure of water hyacinth based on various pretreatment methods. BioResources, 2019, 14 (3): 6088-6099.
CrossRef - Boontum, S; Phetsom, J; Rodiahwati, W; Kitsubthawee, K; Kuntothan T. Characterization of diluted-acid pretreatment of water hyacinth. Applied Science and Engineering Progress, 2019, 12 (4), 253-263.
CrossRef - Biswas B, Banik A.J, Biswas A.B. Comparative study of various pre-treatment techniques for saccharifications of water hyacinth (eichhornia crassipes) cellulose. International Journal of Biotech Trends and Technology (IJBTT), 2015, 5 (3): 11-16.
- Abral, H.; Dalimunthe, M.H.; Hartono, J.; Efendi, R.P.; Asrofi, M.; Sugiarti, E.; Sapuan, S.M.; Park, J.W.; Kim, H.J. Characterization of tapioca starch biopolymer composites reinforced with micro scale water hyacinth fibers. Starch-Stärke 2018.
CrossRef - StanislasT, Nafu Y.R, Tagne N.R.S, Mohamat A.A, Mejouyo P. W. H, Tendo J.F, Njeugna E. Impact of wet-drying treatment of raffia and okra fibres on their morphological, physicochemical and mechanical properties. Journal of Natural Fibers. 2023, 20:1, 2176012, DOI: 10.1080/15440478.2023.2176012
CrossRef - AOAC (Association of Offcial Analytical Chemists) Offcial methods of analysis, 16th Ed. Association of Offcial Analytical Chemists, 1995, Washington DC, USA
- TAPPI T264 cm-97. Preparation of wood for chemical analysis. TAPPI Press, Atlanta, GA.
- ASTM D2495, 2007. Standard Test Method for Moisture in Cotton by Oven-Drying
- ASTM D3822/D3822M-14, 2020. Standard Test Method for Tensile Properties of Single Textile Fibres.
- Nurazzi, N.M.; Asyraf, M.R.M.; Rayung, M.; Norrrahim, M.N.F.; Shazleen, S.S.; Rani, M.S.A.; Shafi, A.R.; Aisyah, H.A.; Radzi, M.H.M.; Sabaruddin, F.A.; et al. Thermogravimetric analysis properties of cellulosic natural fiber polymer composites: A review on influence of chemical treatments. Polymers, 2021, 13, 2710. https:// doi.org/10.3390/polym13162710
CrossRef - Yan Yu . Tuhua Zhong . Yan Wu . Yanjun Li . Zhihui Wu . Benhua Fei. Effect of alkali treatment on microstructure and mechanical properties of individual bamboo fibers. Cellulose, 2017, 24:333–347 DOI 10.1007/s10570-016-1116-6
CrossRef - Singh, A; and Bishnoi, N.R.. Comparative study of various pretreatment techniques for ethanol production from water hyacinth. Industrial Crops and Products, 2013, 44, 283-289. DOI: 101016/j.indcrop.2012.11.026
CrossRef - Biond P, Kuttiraja M, Archana M, Janu K.U, Sindhu R, Sukumaran R.K, Pandey A. High temperature pretreatment and hydrolysis of cotton stalk for producing sugars for bioethanol production. Fuel, 2012, 92: 340–345.
CrossRef - Balat M, Balat H, Oz C. Progress in bioethanol processing. Energ.Combust. 2008, 34: 551–573.
CrossRef - Singh J.K, Chaurasia B, Dubey A, Noguea A.M.F, Gupta A, Kothari R, Upadhyaya, P, Kumar A, Hashem A, Alqarawi A.A, Abd Allah E.F. Biological characterization and instrumental analytical comparison of two biorefining pretreatments for water hyacinth (Eichhornia crassipes) biomass hydrolysis. Sustainability 2021, 13, 245. https://doi.org/ 10.3390/su13010245
CrossRef
- Brandt A., Grasvik, J., Hallett, J.P., and Welton, T. Deconstruction of lignocellulose biomass with ionic liquids. Green Chemistry, 2013, 15 (3), 550-583. DOI: 10.1039/C2GC 363640.
CrossRef
- Mukaratirwa-Muchanyereyi N, Kugara J, Zaranyika M.F. Surface composition and surface properties of water hyacinth (Eichhornia crassipes) root biomass: Effect of mineral acid and organic solvent treatment. African Journal of Biotechnology, 2016, 15 (21) : 897-909. DOI: 10.5897/AJB2015.15068 Article Number: C5453DC58592 ISSN 1684-5315
CrossRef
- Park S, Baker J.O, Himme M.E, Parilla P.A, Johnson D.K. Cellulose crystallinity index: measurement techniques and their impact on interpreting cellulase performance. Journal of Biotechnology for Biofuels, 2010, 3:10,1-10, http://www.biotechnologyforbiofuels.com/content/3/1/10
CrossRef - Motaleb, K. Z. M. A, Brigita, A., & Rimvydas, M. Prospective Utilization of Water Hyacinth and Sugarcane Plant Wastes into a Promising Nonwoven Biomaterial: Development and Improvement of their Physico-Mechanical Properties, 2023. https://doi.org/10.1007/s10570-023-05066-2.
CrossRef - Vijay, R., Singaravelu, D. L., Vinod, A., Sanjay, M. R. & Siengchin, S. Characterization of alkali-treated and untreated natural fbres from the stem of Parthenium hysterophorus. J. Nat. Fibres https://doi.org/10.1080/15440478.2019.1612308 (2019).
CrossRef - Balaji, A. N., & Nagarajan, K. J. Characterization of Alkali Treated and Untreated new Cellulosic Fibre from Saharan Aloe Vera Cactus Leaves. Carbohydates Polymers 174: 200 – 208. Elsevier.
CrossRef - Sumrith, N., Techawinyutham L., Sanjay, M. R. (2020) Characterization of Alkaline and Silane treated fibers of ‘Water Hyacinth Plants’ and Reinforcement of ‘Water Hyacinth Fibres with Bio Epoxy to Develop Fully Biobased Sustainable Ecofriendly Composites. Journal of Polymers and the Environment, 28(10):2749 – 2760. https:// doi. org/ 10. 1007/ S10924- 020- 01810-Y.
CrossRef - Janoobi M, Harun J, Shakeri A, Misra M, Oksman K. Chemical composition, crystallinity, and thermal degradation of bleached and unbleached kenaf bast. Bioresources, 2009, 4: 626–639.
CrossRef
- Gurukarthik Babu, D. Prince Winston, P. Senthamarai Kannan, S.S. Saravanakumar, & M. R. Sanjay (2018). “Study on characterization and physicochemical properties of new natural fiber from Phaseolus vulgaris.” Journal of Natural Fibers, 16 (7): 1035-1042, https://doi.org/10.1080/ 15440478.2018.1448318
CrossRef - Babu B.G, Winston D.P, Kannan P.S, Saravanakumar S.S, Sanjay M.R. Study on characterization and physicochemical properties of new natural fiber from Phaseolus vulgaris. Journal of Natural Fibers, 2018, 16 (7): 1035-1042, https://doi.org/10.1080/15440478.2018.1448318
CrossRef - Arthanarieswaran V.P, Arumugam K, Saravanakumar S.S. Characterization of new natural cellulosic fiber from acacia leucophloea bark. International Journal of Polymer Analysis and Characterization, 2015, 20(4):150311131956002 DOI: 10.1080/1023666X.2015.1018737.
CrossRef
- Chandraseker M, Ishak M.R, Sapuan S.M, Leman Z, Jawaid M. A review on the characterization of natural fibres and their composites after alkali treatment and water absorption. Plastic Rubber Composites, 2017, 46: 119 – 136.
CrossRef - Vinod A, Vijay R, Singaravvelu D. L Sanjay M. R, Siengchin S, Yagnaraj, Y. & Khan, S.M. Natural fibre from stem of Cardiospermum halicababum. Journal of Natural fibres, 2019, 18, 898 – 908.
CrossRef - Bakri, M.K.B, Jayamani E. Comparative study of functional groups in natural fibers: Fourier transform infrared analysis (FTIR). International Conference on Futuristic Trends in Engineering, Science, Humanities, and Technology (FTESHT-16) ISBN: 978-93-85225-55-0, January 23-24, 2016, Gwalior: 167-174.
- Vishnu Vardhini, V.K. J, Murugan, R, Rathinamoorthy, R. Effect of alkali treatment on physical properties of banana fibre. Indian Journal of fibre & Textile Research, 2019, 44: 459 – 465.
- Reddy N, Yang Y. Innovative Biobers from Renewable Resources. Berlin: Book, Springer.
- Tumolva T, Ortenero J, Kubouchi M. Characterization and treatment of water hyacinth fibres for NFRP Composites. In Proceedings of the XIX International Conference of Composite Materials, Canadian Association for Composite Structure and Materials. Montreal, QC, Canada, 28 July–2 August 2013: 1–11
- Meon M S, Othman M F, Husain H, Remeli M F, Syawal M S M. Improving tensile properties of kenaf fibres treated with sodium hydroxide” Procedia Engineering, 2012, 41: 1587–1592.
CrossRef
Abbreviations
WHF: Water hyacinth fibre
XRD: X-ray diffraction analysis
SEM: Scanning electron microscope
TGA: Thermogravimetric Analysis
DTG: Differential Thermogravimetric Analysis
DTA: Differential Thermal Analysis
FTIR: Fourier-Transform Infrared Spectroscopy











