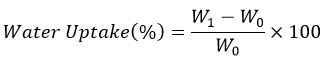Optimization of Natural Rubber and PMMA Grafted Natural Rubber Contents in Soy Protein Isolate Film and Its Characterization
1Department of Biotechnology, Central University of South Bihar, Gaya-, India.
2Department of Materials Science and Engineering, Indian Institute of Technology, Patna, India.
Corresponding Author E-mail: krrakesh72@gmail.com
DOI : http://dx.doi.org/10.13005/msri/220304
ABSTRACT:The growing demand for biodegradable alternatives to synthetic plastics has shifted attention to protein-based bioplastics. This study focused on developing and characterizing films made from soy protein isolate (SPI) blended with natural rubber (NR) and polymethyl methacrylate grafted natural rubber (PMMA-g-NR), with and without a curing agent (CA). The ideal curing time for NR was found to be 30 h at 60°C, resulting in films that are non-sticky and uniform. Visual assessments reveal that all films have semi-transparent, glossy surfaces, with minor color alterations from heat exposure. Fourier Transform Infrared (FTIR) analysis confirmed successful interactions between SPI and both additives, enhanced cross-linking, and network formation in the sample treated with the curing agent. UV-Visible spectroscopy demonstrated that PMMA-g-NR incorporated SPI films have better transparency than NR films, especially when cross-linked with a curing agent. NR-incorporated films clearly outperform PMMA-g-NR films in mechanical properties, highlighting NR's effectiveness in enhancing durability and performance. Water uptake studies revealed that NR increased the hydrophilicity of the films, whereas PMMA-g-NR with curing agent significantly reduced water absorption, i.e., PC25 showed the lowest water uptake (~70%). Additionally, Water Vapor Transmission Rate (WVTR) analysis indicated that the incorporation of additives (especially with curing agent) enhanced barrier properties, and NC25 and PC25 showed the lowest permeability. However, antibacterial assays showed no inhibitory effect against Escherechia coli and Listeria monocytogenes. In summary, the findings indicate that PMMA-g-NR, particularly with a curing agent, outperforms NR in improving the physical and barrier properties of SPI films. This makes them suitable as eco-friendly packaging options.
KEYWORDS:FTIR; Natural rubber; Poly (methyl methacrylate)-grafted natural rubber; Soy protein isolate; Tensile strength.
Introduction
The current era is referred to as the “plastic age” due to the widespread use of synthetic plastics such as polyethylene, polypropylene, and polyvinyl chloride for various purposes.1 Excessive production, consumption, and inappropriate disposal of synthetic plastics have led to widespread plastic pollution and severe environmental effects. Around the globe, environmentalists and governments are becoming quite concerned about this issue.2,3 Petroleum is the main raw material required for producing synthetic plastics, and its depletion releases carbon dioxide and other harmful substances during production, posing a significant risk to the environment and the entire globe. These plastics exist in the environment for hundreds of years despite being discarded because they do not break down naturally3,4 and also the non-disposable nature contaminates the ocean.5
Biodegradable polymers derived from renewable biomass are known as bioplastics. Wood fibers, vegetable lipids, starch or lignin polymers, and other elements are the fundamental building blocks of bioplastics. The majority of biodegradable bioplastics decompose substantially, with little or no harmful substances produced.6 Bioplastics are regarded as being extremely safe for human health and the environment because of their biodegradability.7 Sustainable resources include fibres derived from pineapple, jute, hemp, and banana stems, as well as potato and corn starches,8 newspaper pulp,9 waste paper,10 citrus trash,11 Prosodies juliflora,12 cyanobacteria,13 Pseudomonas putida,14,15 Bacillus sp.,16 and cassava.17 In recent years, there has been a strong push for the use of innovative bioplastics production processes that reduce plastic waste and support sustainable solutions.18
Biodegradable plastics have a bright future compared to traditional plastics, because bioplastics may have a significantly less carbon impact. Apart from carbon efficiency, bioplastics are energy-efficient and less vulnerable to changes in the price of petroleum-based resources since they take less energy to produce.19,20 Protein-based films are under consideration worldwide as a substitute for synthetic polymers because of their accessibility, eco-friendly nature, and superior film-forming capabilities.21 Bioplastics derived from soy protein are altered in a variety of ways to create edible coatings, films, and packaging. Soy protein is derived from the soybean crop, which is primarily cultivated for its oil and protein and has industrial significance.22,23 Soybeans are generally consumed in East Asia as fermented foods like miso, soy sauce, and unfermented foods like boiled soybeans, tofu, and tofu skin. After extracting oil from soybean seeds, the remaining protein components are treated as waste.24 These wastes have the potential to be used in alternative edible packaging, as they have rich protein content and different globulin fractions having film-forming abilities.
Globulins are the main storage protein found in soybeans. Four protein fractions, such as 2S, 7S, 11S, and 15S, are classified according to their sedimentation properties. According to reports, the proportion of 2S, 7S, 11S, and 15S in the total protein composition is 8%, 35%, 52%, and 5%, respectively.25 In comparison to films made from other plant-derived proteins, soy protein-based films are more flexible, smoother, and clearer. The hydrophilic and hydrophobic amino acids that make up soy protein contain various functional groups, allowing interaction with a wide range of additives.26,27
Solution casting and compression molding are two primary methods used to make SPI-based polymers. Bioplastics made from soy protein lack antimicrobial qualities and mechanical strength. However, by adding appropriate additives like licorice extract,28 organic acids, phenolic acids,29 polyphenolic-rich extracts like grape seed,30 and several carboxylic acid compounds, the material characteristics of films can be significantly enhanced. These studies have reported improvements to some extent in material properties like tensile strength, water resistance, and antibacterial properties, which are crucial for developing biopolymeric films. This study hypothesizes that incorporating natural rubber (NR) and Poly (methyl methacrylate) grafted natural rubber (PMMA-g-NR) can enhance the material and functional properties of SPI films.
Natural rubber is a cis-1,4-polyisoprene polymeric material extracted from milky latex produced by Hevea brasiliensis trees. Because of its exceptional elasticity, tensile strength, and resilience, natural rubber is more economically significant in many types of consumer and industrial uses.31 Poly (methyl methacrylate) (PMMA) is a synthetic polymer that originates from methyl methacrylate (MMA) monomer. PMMA works as an amorphous thermoplastic material that displays a colorless appearance and has glass transition temperatures between 100-130 °C while possessing density values of 1.18-1.20 g/cm³ during room temperature conditions. It is characterized by its hydrolysis,32 solubility,33,34 thermal decomposition,35 and other reactive properties. Because of these characteristics, it can be used in high-tech fields such as nanotechnology, optics, and biomedical devices,36 and energy systems, making it an essential component of advanced manufacturing.37 PMMA-g-NR confirms a major innovation in polymer addition, improving NR functional properties by chemical grafting.38
Materials and Methods
Materials
For the preparation of soy protein isolate (SPI) films, SPI powder containing 90% protein was used, which was procured from Pro Foods, India, which is a trademark brand of ChemKart. Glycerol was purchased from Thermo Fisher Scientific India Pvt. Ltd. Sodium hydroxide (NaOH) pellets and sodium chloride (NaCl) were acquired from HiMedia Laboratories Pvt. Ltd., and fused calcium chloride (fused CaCl2) was procured from Sisco Research Laboratories Pvt. Ltd. NR and PMMA-g-NR were used as additional additives sourced from IIT Patna.
Preparation of Soy Protein Isolate Films
The SPI films are formed using the solution casting method. For the preparation of SPI suspension, (7% w/w) of SPI powder is used. The entire process follows these specific procedures. First, a mixture was created by combining 50 ml of distilled water with 1.05 g of glycerol, which is 30% w/w of SPI. The solution was then stirred at 60°C for 15 min. Next, 3.5 g of SPI (7% w/w) was added while stirring continuously, and the pH was maintained at 9.5 using NaOH. After 30 min, the solution is removed from the stirring condition and kept aside for 5-10 min to cool down. Then, for the bubble removal process, the solution was placed under a vacuum desiccator for at least 6 h. In the last step, the final suspension was poured onto a glass petri plate having a coating of silicon, then it was placed under a 60W bulb for 3 h without any disturbance, then left overnight under a 40W bulb to generate the temperature of around 60°C and 40°C, respectively. After this, the petriplate was placed at 75% relative humidity for 24 h to easily peel off the films formed.
Preparation of NR and PMMA-g-NR Incorporated Soy Protein Isolate Films
The same solution casting procedure was followed to cast the composite films as described in the above section. Here, after the addition of SPI followed by stirring for 30 min, additives of different concentrations were added to it. Solid content of NR and PMMA-g-NR was calculated and was found to be 61.03% and 59.92%, respectively. Based on their solid content, NR and PMMA-g-NR of different formulations w/w of SPI (5%, 10%, 15%, 20%, 25%) were added to it and then stirred for an additional 30 min at 60°C. After cooling, the solution was placed in a vacuum desiccator to eliminate any trapped air bubbles. Finally, the bubble-free solution was cast onto a glass petri plate pre-coated with silicone spray and dried with the temperature being maintained at 60°C and 40°C subsequently. The following day, the resulting film was conditioned at 75% relative humidity (RH) for 24 h to ensure easy removal of films from the petri plate. The designation of the NR and PMMA-g-NR incorporated SPI films is given in Table 1.
Table 1: Sample designation of NR and PMMA-g-NR samples
|
Sample designation |
Contents of NR without curing agent (NC) |
Contents of NR with curing agent (NW) |
Contents of PMMA-g-NR without curing agent (PC) |
Contents of NR without curing agent (PW) |
|
With respect to SPI |
With respect to SPI |
|||
|
NC5/ NW5 |
5 |
5 |
– |
– |
|
NC15/ NW15 |
15 |
15 |
– |
– |
|
NC25/ NW25 |
25 |
25 |
– |
– |
|
PC5/ PW5 |
– |
– |
5 |
5 |
|
PC15/ PW15 |
– |
– |
15 |
15 |
|
PC25/ PW25 |
– |
– |
25 |
25 |
Characterization
Transmittance
Transmittance indicates the percentage of light that passes through a material without being altered or absorbed. The optical transmittance of the NR and PMMA-g-NR incorporated SPI films was measured using a UV-VIS Spectrophotometer (UV-1900I SHIMADZU, Japan). For the transmittance analysis, the SPI films were cut into strips measuring 4 cm x 1 cm. Air served as a blank reference during measurement. The film strips were positioned in a cuvette, and transmittance measurements were recorded over a wavelength range from 800 nm to 200 nm.
Mechanical Properties
Tensile strength of the SPI films was prepared using a Universal Testing Machine (Zwick, Germany) at IIT Patna, following the ASTM D882 standard. Film specimens were prepared with dimensions of 8 cm x 1 cm and a thickness of 0.3 mm. Tensile strength and elongation at break were assessed for both NR and PMMA-g-NR integrated SPI films. A crosshead speed of 20 mm/min was used for the testing. Three samples were examined for each film formulation, and the average results are given.
FTIR
FTIR spectroscopy was performed using a Jasco FTIR 4600 spectrophotometer to analyse both NR and PMMA-g-NR incorporated SPI films. The film samples were cut into 1 cm x 1 cm squares and scanned at room temperature. To ensure spectral precision, an average of 32 scans per sample was captured during the 4000–600 cm-1 spectrum collection.
Water Uptake
Water uptake studies were conducted following ASTM D570 to evaluate the relative water absorption of the SPI films. Samples were cut into 1 cm x 1 cm square pieces and kept at 60°C to remove extra moisture absorbed by the film. Following this, all samples were kept at 0% relative humidity (RH) in a dessicator to ensure uniform initial moisture content. The initial dry weight of each sample (W0) of each specimen was recorded. After that, the samples were immersed in 20 ml of distilled water for 24 h. Following soaking, the final weight (W1) was obtained after the surface water was removed with tissue paper. The following formula was used to determine the percentage of water uptake:
Water Vapour Transmission Rate
The amount of water vapor that may flow through a material in a day is measured by the water vapor transmission rate (WVTR). For this, we use a standard water vapour permeability cup. The cup was first filled with 7g of fused calcium chloride, and then an SPI film was used to seal the cup (5.4 cm outer diameter, 2.7 cm inner diameter). Then, the initial weight of the cup was recorded as (W0), and then placed in a dessicator maintained at 75% relative humidity (RH). After 24 h, we took the final weight (W1). The weight change (G = W1 – W0) corresponds to the amount of water vapour transmitted and was calculated using the formula:
G = Final weight (W1) – Initial weight (W0)
Where t = the time in h
A = is the surface area that is taken in m2
Antibacterial Activity
The antibacterial property of both NR and PMMA-g-NR incorporated SPI films was tested using the disc diffusion method. After being cut into 1 cm × 1 cm squares, the film samples were sterilized for 30 min under UV light. Bacterial strains Escherichia coli and Listeria monocytogenes were individually cultured overnight in Luria Broth (LB) medium at 37°C to generate primary cultures. From these, 100μl of secondary culture with an optical density (OD) of 0.05 was evenly spread onto Mueller-Hinton Agar (MHA) plates. The sterilized SPI films were carefully placed onto the bacteria-spread surfaces. After that, the plates were incubated for 24 hours at 37°C. Zones of inhibition surrounding the films were used to measure the antibacterial activity following incubation.
Results
Optimization of Curing Time of NR
Optimization of an experimental design is a need to achieve the best possible results and to understand the variables better. The curing process, better known as vulcanization, is a chemical cross-linking process between the polymer chains and curing agents like sulfur, which in turn increases its material properties like elasticity and durability. At first, the curing time of NR and PMMA-g-NR (with curing agent and without curing agent) was optimized at 60oC for varying durations. It has been observed that at 12 h, films are sticky in nature. At 18 h and 24 h, partial improvements were noted, but the films were still soft. Further, it has been noticed that at 30 h and above, the films were completely non-sticky and dark yellow in color, which suggests that 30 h at 60°C is the optimum curing time for experiments.
Visual Appearance of Films
Upon visual inspection, it has been observed that the color of the NR and PMMA-g-NR incorporated SPI films slightly changed from pale yellow to honey brown after curing, with a smooth, semi-transparent, and glossy surface.
FTIR
FTIR analysis was used to examine the chemical interactions and structural changes in the SPI-based films following the addition of NR or PMMA-g-NR, with and without curing agent. Figure 1 shows the FTIR spectra. Native SPI films displayed typical adsorption peaks such as broad O-H stretching and N-H bending (3000~3500 cm-1) and C-H stretching (2930 cm-1). Other peaks at 1640 cm-1, 1539 cm-1, 1239 cm-1, and 1080 cm-1 were attributed to C=O stretching (amide I), N-H bending (amide II), C-N and N-H stretching (amide III), and C-O stretching, respectively.27
In the NR-incorporated SPI films, the characteristic peaks remained the same, but there was notable broadening and reduction in transmittance intensity, especially observed in NW25. Increasing NR concentration disrupted the SPI matrix, possibly due to limited miscibility and the formation of heterogeneous regions. In the curing agent added NR incorporated SPI films, sharper peaks at 1625 cm-1 and 1453 cm-1 in the NC5 and NC25 indicate that the curing agent facilitated stronger bonding interactions between SPI and NR. These findings suggest improved cross-linking efficiency in the NC-incorporated films compared to NW films.
For PMMA-g-NR incorporated films, a distinct band at 1734 cm-1 indicates ester carbonyl (C=O) stretching, confirming the successful integration of PMMA-g-NR into the matrix. The intensity of this peak increased with higher PMMA-g-NR concentrations. Additionally, bands near 1438 cm-1, 1062 cm-1, and 922 cm-1 were more prominent in PMMA films, particularly in the PC films. This could be due to cross-linking with the curing agent with the SPI matrix.
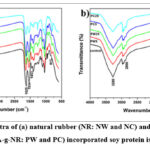 |
Figure 1: FTIR spectra of (a) natural rubber (NR: NW and NC) and (b) PMMA-grafted NR (PMMA-g-NR: PW and PC) incorporated soy protein isolate films.
|
Transmittance
Figure 2 depicts the UV-Visible spectral analysis of NR/PMMA-g-NR incorporated SPI films, revealing notable differences in light transmittance. The SPI control film exhibited the highest transparency across the UV-Vis range, suggesting a homogenous matrix. Upon NR incorporation, particularly at higher concentrations, significantly reduced transmittance and increased film opacity, with or without curing agent, likely due to phase separation. PMMA-g-NR incorporated films exhibited a less significant decrease in transmittance and maintained higher clarity, especially in the presence of the curing agent. All rubber-containing films demonstrated improved UV-blocking capacity compared to the control.
 |
Figure 2: Transmittance spectra of soy protein isolate films incorporated with (a, b) NR (NW and NC) and (c, d) PMMA-g-NR (PW and PC).
|
Mechanical Properties
The mechanical properties of NR and PMMA-g-NR incorporated SPI films are presented in Table 2. The SPI control film showed a tensile strength of 4.82 ± 0.68 MPa and an elongation at break of 181.83 ± 73.44%. Upon analyzing the data, we observed that the pure SPI control film outperforms all modified films in tensile strength. Also, NR-incorporated films show comparatively better mechanical properties than those of PMMA-g-NR-incorporated films. It is also evident that moderate content (15% NR and PMMA-g-NR) has a greater impact on tensile strength compared to 5% and 25%.
Table 2: Mechanical properties of NR and PMMA-g-NR incorporated soy protein isolate films. The data were presented as mean±SD with ‘n’ number of repeats (n = 3).
|
Sample Designation |
Tensile Strength (MPa) |
Elongation at break (%) |
|
Control |
4.82 ± 0.68 |
181.83 ± 73.44 |
|
NC5 |
3.12 ± 0.10 |
24.81 ± 8.01 |
|
NC15 |
3.04 ± 0.19 |
205.06 ± 44.99 |
|
NC25 |
3.67 ± 0.22 |
178.69 ± 31.09 |
|
NW5 |
1.98 ± 0.50 |
10.17 ± 4.19 |
|
NW15 |
4.30 ± 0.53 |
209.40 ± 54.57 |
|
NW25 |
3.41 ± 0.81 |
198.86 ± 12.47 |
|
PC5 |
3.19 ± 0.17 |
30.73 ± 8.91 |
|
PC15 |
1.50 ± 0.24 |
23.67 ± 5.51 |
|
PC25 |
1.79 ± 0.70 |
17.00 ± 5.66 |
|
PW5 |
1.42 ± 0.26 |
16.33 ± 5.69 |
|
PW15 |
3.97 ± 0.37 |
217.06 ± 36.13 |
|
PW25 |
2.06 ± 0.57 |
85.40 ± 57.12 |
Water Uptake
The control SPI film exhibited a water uptake of approximately 120%, as shown in Figure 3. NR-incorporated films without curing agent showed a concentration-dependent increase in water uptake, peaking at NW-25 (~260%), indicating greater matrix swelling. In contrast, NR films with curing agent exhibited relatively lower uptake at higher concentrations (e.g., NC-25) due to enhanced crosslinking and reduced hydrophilicity. PMMA-g-NR based films with or without curing agent showed better water uptake behaviour and were more hydrophobic than the control and NR films. PC25 showed the lowest water absorption (~70%), suggesting improved water resistance due to the synergistic effects of PMMA-g-NR and curing agent. PC5 and PC10 were delicate and could not be retrieved in an intact state to perform further analysis.
 |
Figure 3: Water uptake capacity of soy protein isolate films containing NR and PMMA-g-NR. Data represent mean values ± standard deviation (n = 3).
|
Water Vapor Transmission Rate
WVTR of SPI films with varying NR and PMMA-g-NR concentrations over a specific period of 24 h is shown in Figure 4. The control SPI film showed high water vapor permeability (8.85 g/m2/24h), which signifies poor moisture resistance. NR- and PMMA-g-NR-incorporated films showed decreased WVTR values, especially at 25% concentration. Incorporation of 25% NR (with or without curing agent) displays a significantly reduced WVTR of 5.56 g/m2/24h. Similarly, 25% PMMA-g-NR with or without curing agent added films showed reduced permeability values of 5.90 and 5.38 g/m2/24h, respectively. Thus, it confirms enhanced moisture resistance property due to the densified network structure from crosslinking.
Antibacterial Studies
The antibacterial activity of NR and PMMA-g-NR incorporated soy protein isolate films was assessed against Escherichia coli and Listeria monocytogenes by using the agar diffusion method. The findings reveal no visible zone of inhibition around the film samples for either bacterium. This indicates that the film possesses no inherent antibacterial properties under the test conditions.
Discussion
The optimization of curing time was critical to enhance the structural and functional performance of the NR and PMMA-g-NR incorporated SPI films. At 60°C, curing for less than 24 h resulted in sticky nature of the film, indicating incomplete curing, while 30 h led to a completely cured/cross-linked film with a consistent dark yellow coloration. The temperature of less than 60°C was deliberately chosen to avoid denaturation of SPI while enabling gradual vulcanization of NR and PMMA-g-NR. These observations support that a 30 h period at a temperature of 60°C was optimal for balancing crosslinking without degrading the protein matrix.
The visual transformation of SPI films from pale yellow to honey brown, alongside increased surface gloss and semi-transparency, suggests successful incorporation of plasticizer and rubber materials. The addition of NR and PMMA-g-NR appears to induce thermally driven changes in the film matrix, influencing both color and texture.
FTIR spectra further confirmed structural integration between SPI and the rubber components. The presence of broadened and shifted amide peaks, particularly in films with curing agents, suggests enhanced hydrogen bonding and potential covalent interactions due to crosslinking. For instance, sharper bands at 1625 cm-1 and 1453 cm-1 in cured NR films imply stronger interfacial interactions, likely due to sulfur crosslinking between NR and reactive SPI groups. Similarly, the emergence of a 1734 cm-1 ester carbonyl peak in PMMA-g-NR films confirms successful grafting and its integration into the SPI matrix.
The UV-Vis transmittance data indicated that PMMA-g-NR incorporated films maintained better clarity than NR-only films, especially at higher loadings. This could be attributed to better compatibility and dispersion of PMMA chains in the protein network. The improved UV resistance of all rubber-incorporated films also points to the light-scattering effect of embedded rubber domains, which enhances protection against photo-degradation, a feature particularly desirable in biodegradable packaging films.
Mechanical testing revealed that control films exhibited superior tensile strength and elongation at break compared to all NR and PMMA-g-NR incorporated films. The possible reason could be that the different polarity characteristics of SPI (hydrophilic) and natural rubber (hydrophobic), lead to poor miscibility and phase separation. The possible reason for the negative effect of the curing agent on tensile strength is that curing agents may crosslink with the rubber phase too early during processing, which can cause improper miscibility. It has been observed that the films with 5% and 25% NR and PMMA-g-NR show lower mechanical properties compared to those with 15%. This is likely due to low rubber concentrations forming isolated particles that are poorly dispersed. In contrast, the higher concentration of 25% may result in larger phase separation, further affecting the mechanical properties. Future work should aim to optimize emulsifier incorporation, such as adding whey protein isolate and Tween 80, into SPI-NR/PMMA-g-NR films to improve phase compatibility and prevent phase separation.39 Additional studies are needed to assess the effects of these emulsifiers on film morphology, mechanical properties, and barrier performance, and evaluate antimicrobial options and long-term stability for packaging applications.
Water uptake and WVTR results further highlighted the effectiveness of crosslinking in improving moisture barrier properties. Films with higher concentrations of NR or PMMA-g-NR showed significantly reduced water absorption and vapor transmission, suggesting successful reduction in matrix porosity and enhancement in hydrophobicity. The improved water uptake and WVTR properties are due to the hydrophobic nature of NR and PMMA-g-NR. Curing agents create additional crosslinks between polymer chains, resulting in a more compact network structure, which reduces free volume and molecular mobility, making it harder for water molecules to penetrate.
Lastly, the absence of antibacterial activity in both NR and PMMA-g-NR films is not unexpected, as neither component possesses intrinsic antimicrobial properties. Further investigation should focus on incorporating proven antimicrobial agents such as nalidixic acid,40 mandelic acid,41 essential oil monomers,42 and certain nanoparticles into SPI-based films, aiming to impart effective antibacterial properties.
Conclusion
This study successfully demonstrated the development of bioplastics based on NR and PMMA-g-NR incorporated SPI films, both with and without curing agent. The incorporation of these NR additives with curing agents significantly improved the films’ structural, optical, and barrier properties. FTIR and UV-Visible spectral analysis confirmed molecular interactions and improved integration of additives, particularly in cross-linked films. PMMA-g-NR-based films, especially those having 25% of PMMA-g-NR with curing agent (PC25), exhibited superior performance in terms of transparency, water resistance, and water vapor barrier efficiency. Control films showed higher tensile strength than various NR and PMMA-g-NR formulations, with NR incorporated films exhibiting better mechanical properties than those with PMMA-g-NR. Although no antibacterial activity was detected, these preliminary results suggest that SPI films modified with NR or PMMA-g-NR may offer promising biodegradable packaging alternatives, provided that further studies address antimicrobial performance, long-term stability, and processing scalability.
Acknowledgement
We gratefully acknowledge the Department of Biotechnology, Central University of South Bihar, for providing the laboratory facilities and institutional support necessary for this research. We also thank the university’s central instrumentation facility for access to characterization equipment. Additionally, we thank the Department of Materials Science and Engineering, Indian Institute of Technology, Patna, for providing access to the Universal Testing Machine. This work was supported by the Department of Biotechnology, Government of India, under the DBT Fellowship program.
Funding Sources
Author Mr. Dibyankar Barik is grateful to the Department of Biotechnology (DBT), Government of India, for the financial assistance provided in the form of a Senior Research Fellowship grant (Fellow ID: DBT/2022-23/CUSB/2080).
Conflict of Interest
The authors declare no competing conflict of interest.
Data Availability Statement
The manuscript incorporates all datasets produced or examined throughout this research study.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This study did not require any clinical trials. So, this is not applicable to this research paper.
Authors’ Contribution
Dibyankar Barik – Methodology, analysis, and wrote the manuscript
Abhitej Kumar Jha – Data collection
Rakesh Kumar – Conceptualization, Supervision, and final approval of this manuscript
Dinesh Kumar Kotnees – Supervised the work related to the tensile strength
References
- Thompson RC, Swan SH, Moore CJ, Vom Saal FS. Our plastic age. Philos Trans R Soc B Biol Sci. 2009;364(1526):1973-1976.
CrossRef - Dokl M, Copot A, Krajnc D, et al. Global projections of plastic use, end-of-life fate and potential changes in consumption, reduction, recycling and replacement with bioplastics to 2050. Sustain Prod Consum. 2024;51:498-518.
CrossRef - Luckachan GE, Pillai CKS. Biodegradable polymers- a review on recent trends and emerging perspectives. J Polym Environ. 2011;19(3):637-676.
CrossRef - Emadian SM, Onay TT, Demirel B. Biodegradation of bioplastics in natural environments. Waste Manag. 2017;59:526-536.
CrossRef - Picó Y, Barceló D. Analysis and prevention of microplastics pollution in water: Current perspectives and future directions. ACS Omega. 2019;4(4):6709-6719.
CrossRef - Yamada M, Morimitsu S, Hosono E, Yamada T. Preparation of bioplastic using soy protein. Int J Biol Macromol. 2020;149:1077-1083.
CrossRef - Varshini S, Kesavalakshmi M, Prasath RV, Swetha M, Priya PG, Lakshmi DS. A critical review on role of latex in bioplastics. Waste Manag Bull. 2025;3(3):100199.
CrossRef - Shah M, Rajhans S, Pandya HA, Mankad AU. Bioplastic for future: a review then and now. World J Adv Res Rev. 2021;9(2):056-067.
CrossRef - Siracusa V, Rocculi P, Romani S, Rosa MD. Biodegradable polymers for food packaging: a review. Trends Food Sci Technol. 2008;19(12):634-643.
CrossRef - Sudesh K, Iwata T. Sustainability of biobased and biodegradable plastics. Clean Soil, Air, Water. 2008;36(5‐6):433-442.
CrossRef - Navarro YM, Soukup K, Jandová V, Gomez, MM, Solis JL, Cruz JF, Siche R, Solcova O, Cruz GJF. Starch/chitosan/glycerol films produced from low-value biomass: effect of starch source and weight ratio on film properties. J Phys Conf Ser. 2019;1173(1):012008.
CrossRef - Joshi, S., Sharma, U., Goswami G. Bio-plastic from waste newspaper. Interntional J Eng Res Technol. 2014;2(3):24-27.
- Bátori V, Jabbari M, Åkesson D, Lennartsson PR, Taherzadeh MJ, Zamani A. Production of pectin-cellulose biofilms: a new approach for citrus waste recycling. Int J Polym Sci. 2017;1-9.
CrossRef - Kasirajan, S., Umapathy, D., Chandrasekar, C., Aafrin, V., Jenitapeter, M., Udhyasooriyan, L. Preparation of poly(lactic acid) from Prosopis juliflora and incorporation of chitosan for packaging applications. J Biosci Bioeng. 2019;128(3):323-331.
CrossRef - Bhati R. Biodegradable plastics production by cyanobacteria. Biotechnology Products in Everyday Life. 2018;131-143.
CrossRef - Gatea IH, Abbas AS, Abid AG, Halob AA, Maied SK, Abidali AS. Isolation and characterization of Pseudomonas putida producing bioplastic (polyhydroxy alkanoate) from vegetable oil wastes. J Biotechnol. 2018;15(2):469-473.
- Hadisoewignyo L, Kuncoro F, Tjandrawinata RR. Isolation and characterization of Agung banana peel starch from East Java Indonesia. Int Food Res. J. 2017;24(3):1324-1330.
- Alshehrei F. Antimicrobial activity and gas chromatography-mass spectrometry (GC-MS) analysis of Saudi Arabian Ocimum basilicum leaves extracts. J Pure Appl Microbiol. 2019;13(2):897-904.
CrossRef - Souza AC, Benze R, Ferrão ES, Ditchfield C, Coelho ACV, Tadini CC. Cassava starch biodegradable films: Influence of glycerol and clay nanoparticles content on tensile and barrier properties and glass transition temperature. LWT. 2012;46(1):110-117.
CrossRef - Chozhavendhan S, Usha P, Sowmiya GR. A review on bioplastic production -a need to the society. Int J Pharm Sci Rev Res. 2020;62(1):27-32.
- Visakh PM, Nazarenko O. Soy Protein-Based Blends, Composites and Nanocomposites. Wiley; 2017.
CrossRef - Yu J, Chen LXL. The greenhouse gas emissions and fossil energy requirement of bioplastics from cradle to gate of a biomass refinery. Environ Sci Technol. 2008;42(18):6961-6966.
CrossRef - Arikan EB, Ozsoy HD. A review: investigation of bioplastics. J Civ Eng Archit. 2015;9(2):188-192.
CrossRef - El-Shemy H. Soybean: Bio-Active Compounds. IntechOpen; 2013.
CrossRef - Wu J, Sun Q, Huang H, Duan Y, Xiao G, Le T. Enhanced physico-mechanical, barrier and antifungal properties of soy protein isolate film by incorporating both plant-sourced cinnamaldehyde and facile synthesized zinc oxide nanosheets. Colloids Surf., B.2019;180:31-38.
CrossRef - Nagano T, Hirotsuka M, Mori H, Kohyama K, Nishinari K. Dynamic viscoelastic study on the gelation of 7S globulin from soybeans. J Agric Food Chem. 1992;40(6):941-944.
CrossRef - Dawson P, Hirt D, Rieck J, Acton J, Sotthibandhu A. Nisin release from films is affected by both protein type and film-forming method. Food Res Int. 2003;36(9-10):959-968.
CrossRef - Han Y, Yu M, Wang L. Preparation and characterization of antioxidant soy protein isolate films incorporating licorice residue extract. Food Hydrocoll. 2018;75:13-21.
CrossRef - Insaward A, Duangmal K, Mahawanich T. Mechanical, optical, and barrier properties of soy protein film as affected by phenolic acid addition. J Agric Food Chem. 2015;63(43):9421-9426.
CrossRef - Sivarooban T, Hettiarachchy NS, Johnson MG. Physical and antimicrobial properties of grape seed extract, nisin, and EDTA incorporated soy protein edible films. Food Res Int. 2008;41(8):781-785.
CrossRef - Blackley DC. Polymer Latices. Springer Netherlands; 1997.
CrossRef - Subramaniam A. Natural Rubber. In: Rubber Technology. Vol 61. Springer US; 1987:179-208.
CrossRef - Ishitake K, Satoh K, Kamigaito M, Okamoto Y. From-syndiotactic-to-isotactic stereogradient methacrylic polymers by RAFT copolymerization of methacrylic acid and its bulky esters. Polym Chem. 2012;3(7):1750-1757.
CrossRef - Ueno K, Fukai T, Nagatsuka T, Yasuda T, Watanabe M. Solubility of poly(methyl methacrylate) in ionic liquids in relation to solvent parameters. Langmuir. 2014;30(11):3228-3235.
CrossRef - Evchuk IY, Musii RI, Makitra RG, Pristanskii RE. Solubility of polymethyl methacrylate in organic solvents. Russ J Appl Chem. 2005;78(10):1576-1580.
CrossRef - Van Krevelen DW. Properties of Polymers: Their Correlation with Chemical Structure; Their Numerical Estimation and Prediction from Additive Group Contributions: Fourth Edition. Elsevier; 2009.
- Nien YH, Lin SW, Hsu YN. Preparation and characterization of acrylic bone cement with high drug release. Mater Sci Eng C. 2013;33(2):974-978.
CrossRef - Saramolee P, Lopattananon N, Sahakaro K. Preparation and some properties of modified natural rubber bearing grafted poly(methyl methacrylate) and epoxide groups. Eur Polym J. 2014;56(1):1-10.
CrossRef - Yang T, Liu C, Zheng Y, Liu TC, Li K, Liu J, Liu Y, Zhou P. Effect of WPI/Tween 80 mixed emulsifiers on physicochemical stability of ginsenosides nanoemulsions. Food Biosci. 2023;53:102519.
CrossRef - Rani S, Singh AK, Paswan RR, Kumar KD, Kumar R. Preparation, characterization and antibacterial evaluation of soy protein isolate biopolymeric films loaded with nalidixic acid. J Polym Environ. 2020;28(7):1841-1850.
CrossRef - Kumar R, Rani P, Kumar KD. Soy protein isolate film by incorporating mandelic acid as well as through fermentation mediated by Bacillus subtilis. J Renew Mater. 2019;7(2):103-115.
CrossRef - Zhang Y, Jiang J. Physical properties and antimicrobial activities of soy protein isolate edible films incorporated with essential oil monomers. Adv. Mater. Res. 2012;361-367.
CrossRef